LITHIUM ISOTOPES
Enriching the Future
of Energy, One Atom
at a Time

Enriching the Future
of Energy, One Atom
at a Time

Powering Breakthroughs
Across Industries
On-site lithium energy storage enables data centers to smooth peak demand, stabilize local grids, and operate as active participants in regional power systems rather than passive consumers.
Revolutionizing Isotope Separation
EnergyX is developing a proprietary electrochemical and membrane-based enrichment process for precision separation at scale. The system is energy-efficient and modular, suitable for both fusion and battery-grade production. Provisional patents cover multiple separation pathways now in validation.

Understanding Lithium Isotopes
Lithium occurs naturally as two stable isotopes: Li-6 (7.5%) and Li-7 (92.5%). While they share chemical properties, their atomic composition makes each essential for specific energy applications. EnergyX is creating a clean, scalable enrichment process that eliminates toxic reagents and reduces costs compared to legacy chemical separation methods.
Lithium 6
Lithium 6 is a stable isotope making up ~7.59% of the world’s natural lithium. It contains 3 protons, 3 neutrons, and 3 electrons, with an atomic mass of 6.015. Lithium-6 is special, because it has an extremely strong reaction when bombarded with neutrons. While stable, lithium-6 is like a spring-loaded molecular complex. Its nucleus readily absorbs a neutron, goes into a very high energy state, and immediately decays to make helium, tritium, and heat. Tritium is the workhorse for nuclear fusion. Tritium is radioactive and difficult to store safely, making lithium-6 an essential, stable, and safe feedstock for the generation of tritium (“tritium breeding”). These special properties, while critically important for fusion reactors and tritium breeding, are the exact reason isotopically pure Lithium-7 is needed for nuclear fission reactors.
Quick Facts
Atomic Number
3
Primary Use
Tritium production, fusion, advanced reactor
Atomic Weight
6.015 / 7.016
Processing Need
High-precision isotope separation
Lithium 7
Lithium-7 is the predominant stable isotope, making up ~92.41% of the world’s natural lithium. It contains 3 protons, 4 neutrons, and 3 electrons, with an atomic mass of 7.016. Opposite of its companion Lithium-6, Lithium-7 is a neutron-benign atom that enables safe, efficient uranium fission by suppressing tritium production and preserving neutron economy. During nuclear fission in a traditional uranium-fueled plant, corrosive acid is generated in the cooling water as a byproduct of the reaction, corroding and destroying the fuel cladding surfaces and steam generators. To prevent this, 7-LiOH (Lithium-7 hydroxide) is added to the cooling water to control the pH and consume the corrosive acid.
Quick Facts
Atomic Number
92
Primary Use
Fuel
Atomic Weight
238.03
Energy Density
High Density

LITHIUM-6 vs LITHIUM-7
Lithium-6 (⁶Li)
Property
Lithium-7 (⁷Li)
~6.015 amu
Atomic Mass
~7.016 amu
~7.5% of natural lithium
Natural Abundance
~92.5% of natural lithium
Strong neutron absorber
Neutron Behavior
Essentially neutron transparent
Tritium production for fusion and nuclear weapons
Primary Role in Nuclear
Reactor coolant/moderator chemistry and structural material compatibility
⁶Li + n → ³H (tritium) + ⁴He
Key Reaction
Stable under neutron flux, minimal activation
Critical for fusion energy and national security applications
Strategic Importance
Essential for safe and efficient operation of fission reactors
Fusion R&D, isotope research
Civilian / Industrial Uses
Traditional uranium powered n
Highly controlled, limited global producers
Supply Sensitivity
limited global producers
Heavily regulated and export-controlled
Regulatory Status
Lightly regulated outside of nuclear use
Enables future fusion fuel cycles
Energy Relevance
Supports current nuclear fission infrastructure
Powering the Next Generation of Fusion and Advanced Energy Systems
Global Li-6 and Li-7 demand is projected to exceed $10B by 2035, driven by fusion development, and reactor modernization.

Fusion Energy
Li-6 is essential for producing tritium fuel in deuterium-tritium fusion reactions.

Fission & Reactor Cooling
Li-7 is used in reactor coolant systems where low neutron absorption is critical for stability and performance.


How a tomahawk fission reactor Works
1. Vacuum Vessel
A huge stainless steel container that holds plasma and houses the fusion reaction.
2. heating
Neutral beam injections and RF electromagnetic waves heat plasma to 150,000,000 oC.
3. magnets
10k Tons of superconducting magnets generate 200k times that of earth’s magnetic field that confine and shape the plasma.
4. blanket
4 Ton tiles protect the vacuum vessel and magnets from heat and neutrons.
5. divertor
A series of tungsten tiles under the vacuum vessel divert heat and gases from the reactor.
6. diagnostics
Tools Constantly measuring the physics of the plasma generation.
7. cryostat
Refrigeration that surrounds the vacuum vessel, protecting the superconducting magnets and other equipment from heat.
Current and Projected Lithium 6 & 7 Demand
Over the next decade, demand for enriched ⁷Li is projected to increase from less than 100 metric tons per year today to more than 1,000 metric tons per year by 2035, driven by molten-salt reactor development and nuclear-coolant salt production.
Enriched ⁶Li demand, though smaller, is expected to reach tens of metric tons per year to support tritium breeding in fusion demonstrators such as ITER and commercial ventures including Commonwealth Fusion Systems and Helion Energy.
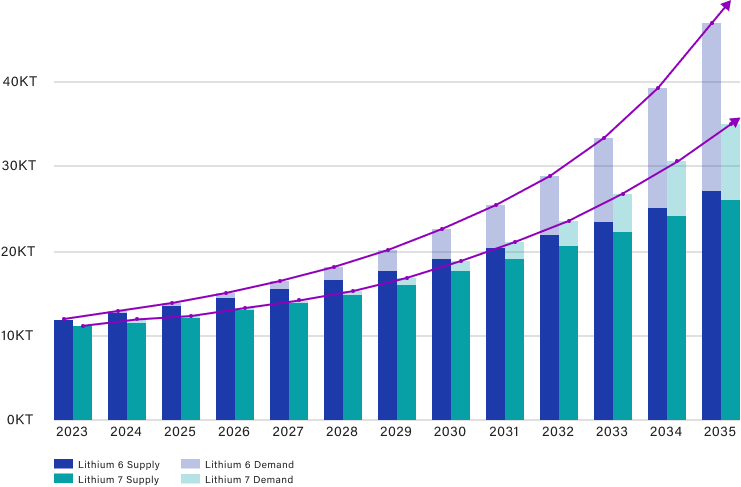

A Critical Supply Gap and a Multi-Billion-Dollar Opportunity
Global Li-6 and Li-7 demand is projected to exceed $10B by 2035, driven by fusion development, and reactor modernization. The fusion segment alone is expected to grow at over 40% CAGR through 2035, with supply lagging far behind demand.
Get in touch
Reach out, and let’s create the future of energy together!
